
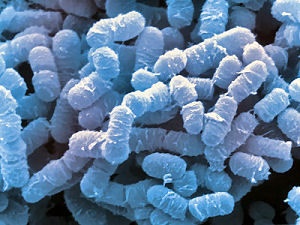
Microbes may be smarter than we think, at least that’s according to Princeton University researchers who have shown for the first time that bacteria don’t just react to changes in their surroundings – they anticipate and prepare for them. The findings, reported in Science, challenge the prevailing notion that only organisms with complex nervous systems have this ability.
The biologists and engineers who conducted the research used lab experiments to demonstrate this phenomenon in common bacteria. They also turned to computer simulations to explain how a microbe species’ internal network of genes and proteins could evolve over time to produce such complex behavior.
“What we have found is the first evidence that bacteria can use sensed cues from their environment to infer future events,” explained Saeed Tavazoie, a molecular biologist who conducted the study along with graduate student Ilias Tagkopoulos and post-doctoral researcher Yir-Chung Liu.
In one part of the research, the team studied the behavior of Escherichia coli (E.coli) to explain a long-standing question about the bug: How do its genes respond to the temperature and oxygen changes that occur when the bacterium enters the gut? The conventional answer is that it reacts to the change – after sensing it – by switching from aerobic (oxygen) to anaerobic (oxygen-less) respiration. If this were true, however, the organism would be at a disadvantage during the time it needed to make the switch. “This kind of reflexive response would not be optimal,” Tavazoie said.
To prove that this was not just genetic coincidence, the researchers then grew the bacteria in a biologically “flipped” environment where oxygen levels rose following an increase in temperature. Remarkably, within a few hundred generations the bugs partially adapted to this new regime, and no longer turned off the genes for aerobic respiration when the temperature rose. “This reprogramming clearly indicates that shutting down aerobic respiration following a temperature increase is not essential to E. coli’s survival,” said Tavazoie. “On the contrary, it appears that the bacterium has “learned” this response by associating specific temperatures with specific oxygen levels over the course of its evolution.”
Lacking a brain or even a primitive nervous system, how is a single-celled bacterium able to pull off this feat? Whereas higher animals can learn new behavior within a single lifetime, bacterial learning takes place over many generations and on an evolutionary time scale, Tavazoie explains.
To gain a deeper understanding of this phenomenon, his team developed a virtual microbial ecosystem, called “Evolution in Variable Environment.” Each microbe in this novel computational framework is represented as a network of interacting genes and proteins. An evolving population of these virtual bugs competes for limited resources within a changing environment, mimicking the behavior of bacteria in the real world. “Simulations at this scale and complexity would have been impossible in the past,” says co-researcher Tagkopoulos. Even with the vast number crunching power the supercomputers provided by the University’s Computational Science and Engineering Support Group, the experiments took nearly 18 months to run.
In the virtual world, microbes are more likely to survive if they conserve energy by mostly turning off the biological processes that allow them to eat. The challenge they face then is to anticipate the arrival of food and turn up their metabolism just in time. To help them along, the researchers gave the bugs cues before feeding them, but the cues had to appear in just the right pattern to indicate that food was on its way. “To predict mealtimes accurately, the microbes would have to solve logic problems,” says Tagkopoulos, the principal architect of the “Evolution in Variable Environment” simulation.
Tagkopoulos said that sure enough, after a few thousand generations, an ecologically fit strain of microbe emerged which did exactly that. This happened for every pattern of cues that the researchers tried. The feeding response of these gastronomically savvy bugs peaked just when food was offered.
When the researchers examined a number of fit virtual bugs, they could at first make little sense out of them. “Their biochemical networks were filled with seemingly unnecessary components,” says Tagkopoulos. “That is not how an engineer would design logic-solving networks.” Pared down to their essential elements, however, the networks revealed a simple and elegant structure. The researchers were able to trace the different sequences of gene and protein interactions the organisms used in order to respond to cues and anticipate mealtimes. “It gave us insights into how simple organisms such as bacteria can process information from the environment to anticipate future events,” says Tagkopoulos.
The researchers say that their findings open up many exciting avenues of research. They are planning to use similar methods to study how bacteria exchange genes with one another (horizontal gene transfer), how tissues and organs develop (morphogenesis), how viral infections spread, and other core problems in biology. “What is really exciting about our discovery is that it brings together and establishes deep connections between the traditionally separate fields of microbial ecology, network evolution, and behavior,” says Tavazoie.
Related:
1st Rule Of Evolution: Strive For Complexity
Biochemical “Noise” Critical For Regulating Biological Processes
Sex Life Of Bacteria Under The Microscope
Bacterial Behaviour Poorly Understood
Horizontal Gene Transfer Vastly Underestimated, Suggests New Study
Sex Optional For Evolutionary Adaptation














Comments are closed.