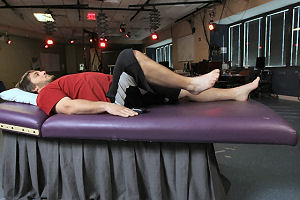
Epidural electrical stimulation, which delivers a continuous electrical current to the lower spinal cord, has allowed four young men who had been paralyzed for years to move their legs. All four participants were classified as suffering from chronic, motor complete spinal cord injuries and were unable to move their lower extremities prior to the implantation of the stimulator. The research, detailed in the journal Brain, was conducted by researchers from the University of Louisville, UCLA, and the Pavlov Institute of Physiology.
The research builds on an initial 2011 study with one subject that found epidural stimulation could recover a number of the patient’s motor functions. Now, three years later, four subjects were able to execute voluntary movements immediately following the implantation and activation of the stimulator.
The four paralyzed participants ranged in neurological level from C7-T5 and were at least two years post-injury at the time of the intervention. Two of them had been rated “A” on the American Spinal Injury Association’s classification system, meaning they had absolutely no sensation or cognition below the site of their injury; the researchers were initially highly sceptical that these men would elicit any voluntary movement as a result of the intervention.
“Two of the four subjects were diagnosed as motor and sensory complete injured with no chance of recovery at all,” said lead researcher Claudia Angeli, from the University of Louisville’s Kentucky Spinal Cord Injury Research Center. “Because of epidural stimulation, they can now voluntarily move their hips, ankles and toes. This is groundbreaking for the entire field and offers a new outlook that the spinal cord, even after a severe injury, has great potential for functional recovery.”
In epidural stimulation, the electrical current is applied at varying frequencies and intensities to specific locations on the lumbosacral spinal cord, corresponding to the dense neural bundles that largely control the movement of the hips, knees, ankles and toes. With the participants, once the signal was triggered, the spinal cord reengaged its neural network to control and direct muscle movements.
“We have uncovered a fundamentally new intervention strategy that can dramatically affect recovery of voluntary movement in individuals with complete paralysis, even years after injury,” said co-researcher Susan Harkema, also from the University of Louisville. “The belief that no recovery is possible and complete paralysis is permanent has been challenged.”
Beyond regaining voluntary movement, Harkema says the research participants have displayed a myriad of improvements in their overall health, including increases in muscle mass and regulation of their blood pressure, as well as reduced fatigue and dramatic improvements to their sense of well-being.
“This research brings up an amazing number of possibilities for how we can develop interventions that will help people recover movement they have lost,” said UCLA’s V. Reggie Edgerton, a distinguished professor of integrative biology and physiology, neurobiology, and neurosurgery and a co-author of the research. “The circuitry in the spinal cord is remarkably resilient. Once you get them up and active, many physiological systems that are intricately connected and that were dormant come back into play.”
The study offers hope that clinical therapies can be developed to advance treatment for the nearly 6 million Americans living with paralysis, including nearly 1.3 million with spinal cord injuries.
“This is a wake-up call for how we see motor complete spinal cord injury,” said Edgerton. “We don’t have to necessarily rely on regrowth of nerves in order to regain function. The fact that we’ve observed this in four out of four people suggests that this is actually a common phenomenon in those diagnosed with complete paralysis.”
Related:
Discuss this article in our forum
Ultrasound used to stimulate tactile sensations in brain
Thought-controlled robotic arm demonstrated
More Success With Stem Cells For Spinal Injuries

















Comments are closed.