Scientists trying to understand the cellular mechanics of how cancer begins have discovered a new type of cell division which they have named klerokinesis. Researcher Mark Burkard, from the University of Wisconsin, reported his team’s findings yesterday at the American Society for Cell Biology Annual Meeting in San Francisco.
Burkard believes that klerokinesis could be an evolutionary failsafe mechanism, the function of which is to “rescue” a range of cell functions during embryonic development. The discovery has major implications for cancer research, according to Burkard, who “hopes to learn how to promote klerokinesis to help prevent cancer.”
Burkard explained that an enormous number of cell divisions occur once a single egg is fertilized (known as mitosis). Replication accuracy during mitosis was an early concern of Theodor Boveri, the German cell biology pioneer who in 1914 published the hypothesis that mistakes that occur when chromosomes divide during the mitotic cell cycle would lead to aneuploidy, characterized by too many or too few chromosomes in the resulting daughter cells. Such abnormal cells, Boveri believed, would lead to the out-of-control cell division that is a hallmark of cancer.
During the century since Boveri’s hypothesis was published, cell biologists have found considerable evidence that aneuploidy can promote the transformation of normal cells into cancer. Until the recent arrival of live single-cell videomicroscopy techniques, it wasn’t possible to examine a key feature of Boveri’s theory: that aneuploidy results from the failure of cytokinesis, the separation of the cell membrane between segregated chromosomes to form two daughter cells.
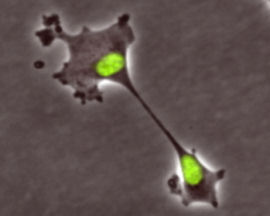
Burkard and his University of Wisconsin colleagues allowed human retinal pigment epithelial (RPE) cells in the lab to undergo karyokinesis, the orderly separation of chromosomes in mitosis, but blocked cytokinesis, the actual division into two daughter cells. This process resulted in single cells with two nuclei, termed binucleate. These binucleate cells were then allowed to go through more mitotic cycles and proliferate.
The researchers said that they were surprised that one-third of the binucleate cells generated healthy daughter colonies. In addition, the majority of their progeny had chromosome sets that perfectly matched the original first-generation RPE cell. “This made Boveri’s random chromosome assortment seem rather improbable,” Burkard explained, “so we carefully observed the cells by time-lapse microscopy to see how this might be occurring.”
They discovered that cells with two nuclei got stuck in the first growth phase of the cell cycle long enough for them to stretch apart, dividing the cytoplasm into two cells, each with its own nucleus. The process neatly preserved the accurately separated chromosome sets. Further work showed that these cells managed to divide without the usual proteins required for division of cell membranes in cytokinesis. Burkard concluded that he was observing a new type of cell division.
He added that the name is derived from the Greek root for allotted inheritance, chosen because each daughter cell inherits a full set of chromosomes. “Klerokinesis is a primitive mechanism of cell division that appears to be preserved in humans, as similar divisions have been observed in organisms such as slime molds,” he noted.
Related:
Discuss this article in our forum
Living cell turned into a laser
Fundamental misunderstanding of cell signaling revealed
Stem cell researchers barking up the wrong tree, says Prof
Synthetic genome successfully transplanted into cell







Comments are closed.