
One of the problems with common photovoltaic cells is that the sun’s rays can be highly destructive to the materials used in the cell, leading to a gradual degradation of its performance. But plants have adopted an interesting strategy to address this issue: They constantly break down their light-capturing molecules and reassemble them from scratch, so the basic structures that capture the sun’s energy are, in effect, always new and at optimum efficiency. Now, MIT scientists have succeeded in mimicking a key aspect of that process.
In Nature Chemistry, lead researcher Michael Strano and his team report how they created a novel set of self-assembling photovoltaic molecules that can be repeatedly broken down and then reassembled quickly.
Strano says the idea first occurred to him when he was reading about plant biology. “I was really impressed by how plant cells have this extremely efficient repair mechanism,” he says. In full summer sunlight, “a leaf on a tree is recycling its proteins about every 45 minutes, even though you might think of it as a static photocell.”
In photosynthesis in plants, the reactive form of oxygen produced by sunlight causes the proteins to fail in a very precise way. As Strano describes it, the oxygen “unsnaps a tether that keeps the protein together,” but the same proteins are quickly reassembled to restart the process.
This action all takes place inside tiny capsules called chloroplasts that reside inside every plant cell – and which is where photosynthesis happens. The chloroplast is “an amazing machine,” Strano says. “They are remarkable engines that consume carbon dioxide and use light to produce glucose,” a chemical that provides energy for metabolism.
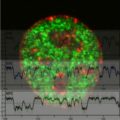
To imitate that process, Strano and his team produced synthetic molecules called phospholipids that form discs; these discs provide structural support for other molecules that actually respond to light, in structures called reaction centers, which release electrons when struck by particles of light.
The system is made up of seven different compounds, including the carbon nanotubes, the phospholipids, and the proteins that make up the reaction centers, which under the right conditions spontaneously assemble themselves into a light-harvesting structure that produces an electric current.
Strano believes this sets a record for the complexity of a self-assembling system. When a surfactant is added to the mix, the seven components all come apart and form a soupy solution. Then, when the researchers removed the surfactant by pushing the solution through a membrane, the compounds spontaneously assembled once again into a perfectly formed, rejuvenated photocell.
The prototype cell ran through repeated cycles of assembly and disassembly over a 14-hour period, with no loss of efficiency. “We’re basically imitating tricks that nature has discovered over millions of years – reversibility, the ability to break apart and reassemble,” Strano said.
Theoretically, the efficiency of the structures could be close to 100 percent, Strano says. But in the initial work, the concentration of the structures in the solution was low, so the overall efficiency of the device – the amount of electricity produced for a given surface area – was very low. The team are now working to find ways to increase the concentration.
Related:
Electricity generated directly from photosynthesis
Quantum Shenanigans Of Photosynthesis Mapped
Robustness of quantum entanglement in photosynthesis surprises researchers
Evolutionary Algorithms Used To Design A Better Leaf
















Comments are closed.