8 August 2006
DNA Structure Still Surprises
by Kate Melville
 Biological dogma has long held that as a molecule of DNA is stretched, its double helix starts to unwind; but Lawrence Berkeley National Laboratory researchers have shown that this is most definitely not the case.
Biological dogma has long held that as a molecule of DNA is stretched, its double helix starts to unwind; but Lawrence Berkeley National Laboratory researchers have shown that this is most definitely not the case.
The experiment, reported in Nature, used microscopic beads and magnetic tweezers to observe that when a DNA molecule is stretched, it actually begins to overwind. This overwinding continues until the force being applied to stretch the DNA exceeds about 30 picoNewtons, at which point the DNA double helix did begin to unwind in accordance with predictions.
"DNA's helical structure implies that twisting and stretching should be coupled, hence the prediction that DNA should unwind when stretched," said Berkeley biophysicist Carlos Bustamante, who led this experiment. "That is why it was such [a] surprise when we directly measured twist-stretch coupling to find instead DNA overwinds when stretched. The DNA molecule, when studied at close range, continues to surprise us!"
The mechanical properties of the DNA double helix are of intense interest to biologists as these properties drive many of the replication and transcription functions in DNA. To get to grips with these characteristics, Bustamante developed a technique called "rotor bead tracking."
In rotor bead tracking, a single DNA molecule is anchored to a surface and a magnetized bead is attached to the free end. A point along the double-helix is then biochemically "nicked" to create a single strand of DNA that acts as a free swivel. Immediately below this nick, a plastic bead is attached to the DNA to serve as a "rotor" that will spin in response to torque. Magnets are used to manipulate the magnetized bead, providing a measured and highly controlled amount of tension to stretch the DNA molecule. With the use of a fluorescent coating, the subsequent spinning of the rotor bead in response to the stretching can be recorded.
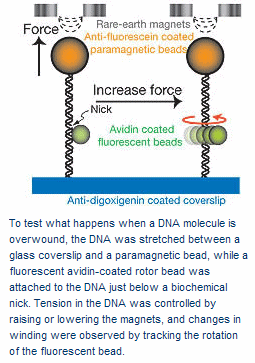 "When we apply tension to the DNA molecule, changes in the rotor bead angle reflect changes in the twist of the lower DNA segment," Bustamante said. "The overwinding observed upon stretching, implies that contrary to the held belief, the stretch-torsion coupling constant of DNA is a negative value. This observation also implies that if we overwind the DNA, the molecule should get longer. Indeed, we found that overwinding caused the DNA molecule to extend by about 0.5 nanometers per turn."
"When we apply tension to the DNA molecule, changes in the rotor bead angle reflect changes in the twist of the lower DNA segment," Bustamante said. "The overwinding observed upon stretching, implies that contrary to the held belief, the stretch-torsion coupling constant of DNA is a negative value. This observation also implies that if we overwind the DNA, the molecule should get longer. Indeed, we found that overwinding caused the DNA molecule to extend by about 0.5 nanometers per turn."
Bustamante has proposed a simple model to explain the overwinding. The model consists of an elastic rod that is wrapped around its outer surface by a stiff wire, analogous to DNA's sugar-phosphate backbone. The elastic rod is constructed from a material that conserves volume under stress. "As this system is stretched, the elastic rod decreases in diameter," said Bustamante. "This enables the outer wire to wrap a larger number of times over the length of the rod." The twist-stretch coupling results demonstrated hold important implications for how DNA-binding proteins are able to recognize their target sites along the helix. These proteins are known to bend, wrap, loop and twist DNA. The new findings show that they can achieve this by simultaneously stretching and overwinding a DNA molecule, or by compressing and underwinding it.
Bustamante said the findings might have application beyond the field of biology. "We believe that our work... will also have implications in nanotechnology. For example, the DNA molecule might provide the energy to power future nanomotors."
Source: Lawrence Berkeley National Laboratory
Graphic courtesy Lawrence Berkeley National Laboratory
