Im amazed at the way you act as if marosz and myself are
somehow wrong about things its almost as if you believe that
by using your cherry picked BS you can somehow correct your
really large pile of wrongness through deception or manipulation
of extremely out of context post.
also: in almost every post in this thread that you have made
you say that you are done with it ... and your not going to
reply any more ... your ignoring me ... or some wording to
that extent.
being wrong seems to be entangled into your every thought
and mental processes this is one reason why I always check
what you write for validity.
as in the below that sam questioned
But I don’t know if a photon tends to become longer in wavelength, with each “re-emission,” after being absorbed, in successive encounters along its mean free path, though I suspect so.
To be clear, I do know it’s not the “same” photon being “re-emitted” after being absorbed, but for the sake of this cartoonlike description, it is the “only” photon we’re following.
I expect that (after being absorbed and ‘re-emitted’) it would be of a somewhat longer wavelength over 80% of the time or maybe even 95% of the time, and only re-emitted at the same frequency less than 20% of the time and more likely less than 5% of the time.
I also wonder if the “re-emitted” photon could be of a somewhat shorter wavelength than the absorbed photon, on rare occasions (probably less than 1% of the time), if a newly energized CO2 molecule (with recently absorbed photon) were to be kinetically bumped with enough force at the right time and angle.
and he then asked
So, do these notions of mine, about how heat changes (usually getting longer) as it undergoes successive radiative transfer, have any meaning or validity or utility? Thanks for your time!
and your reply
Yes all your ideas are all valid, how important or relevant they are to any given pathway will vary and I am not sure you can use them as a general rule.
you just agreed that a range of 80% to 95% of
all photon emissions of a co2 molecule
will have a longer wavelength.
and you also agreed that only a range of 20% to 5% of
all photon emissions of a co2 molecule
will have the same wavelength as the absorbed photon.
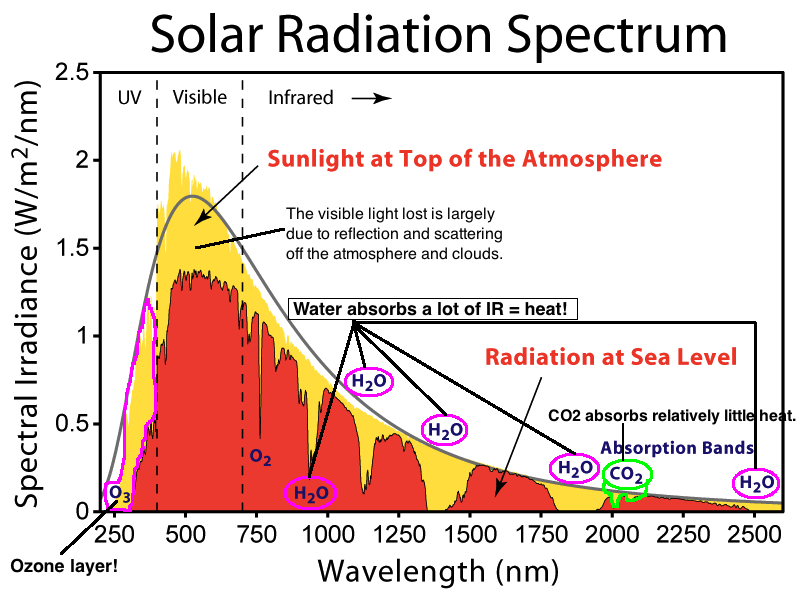
thats really interesting since the infrared value of
carbon dioxide gas is either 667.38 or 2349.16
and its two frequencies are 667 cm-1 or 2350 cm-1
https://en.wikipedia.org/wiki/Carbon_dioxideCarbon dioxide is a greenhouse gas, absorbing and emitting infrared radiation at its two infrared-active vibrational frequencies
this is revealing about both yours and samwiks character
and since samwik has taken classes in climate science or
whatever this fake science that you two are promoting
really shows the extremes that you two will go to in order
to prop up your BELIEFS because this garbage that you guys
are promoting certainly isnt science.